 
Salvarsan, Erlich's "compound 606" (arsphenamine), was the first drug to treat syphilis. White arsenic, arsenic (III) oxide, As4O6, common in sulfide ore deposits, very toxic, rodenticide. In timber treatments, wood preservatives, pesticides, found free and in combined many minerals, three allotropes are yellow, black and main allotrope grey arsenic sublimes at 613 oC, and n-type dopant of silicon semiconductors, hardens lead alloys. So use 5 X the volume of uncooked rice for cooking water and discard the excess water when cooking is finished. Maximum intake of arsenic occurs when the cook only just covers uncooked rice in the pot with water, to be absobed by rice during cooking. High levels of arsenic have been reported in rice grains, particularly unpolished rice. 
Heated arsenic (III) oxide gives off the garlic smell of arsenic and a black ring of arsenic in the test-tube.Īrsenic (III) oxide is amphoteric and is slightly soluble in water.Īrsenic occurs in realgar As4S4, orpiment As2S3, arsenolite As2O3, and arsenopyrite FeAsS.Īrsenic, As, (Greek arsenikon yellow orpiment), arsenic trisulfide, As, metalloid, arsenic mineral, natural arsenic.Īrsenic and arsenic compounds are Not permitted in schools.Ī 65 mg dose or repeated smaller doses may be poisonous.Īlso, small quantities of arsenic may be carcinogenic. When heated it oxidizes to arsenous oxide, which has a garlic odour. It has steel grey colour and is a very brittle, crystalline, semimetallic solid, a metalloid solid. 1.97 and grey arsenic, metallic arsenic S.G. It is used in dyes, pigments, medicines, lead shot alloy, glass-making, fireworks.Īrsenic and arsenic compounds are not use in school science experiments, because these substances are very poisonous. Wood treated with copper chrome arsenate (CCA), 12.2.2.1Īrsenic is widespread and abundant in the earth. Tests for metals with flame tests, 12.11.3.2a Reactions of arsenic and arsenic compounds, 12.2.2 Nickeline, niccolite, arsenical nickel, NiAs, 35.20.30 (Geology) Minerals containing arsenic, 35.20.2.1, (Geology) See 12.11.7.2b, Group IIb As2S3, Sb2S3, SnS, SnS2Īrsenic, light blue (moistened with hydrochloric acid) Tests for arsenic)ġ2.11.7.2 Group II Sulfides insoluble in dilute hydrochloric acid Heat substances with charcoal and fusion mixture, 12.11.3.8 (See, 2. The sodium chloride solution coagulates the control.Īrsine, arsenic hydride, are formed by old green wallpaper. 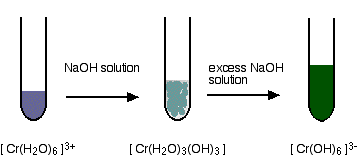
Dilute 2 mL of the antimony sulfide solution with 2 mL of water to act as a control. The sodium chloride solution has no effect where the solution is protected by the starch.Ĥ. Add 2 mL of starch solution to 2 mL of antimony sulfide solution. Please send comments to: elements, reactionsġ2.2.4 Bismuth, Reactions of Bi compoundsġ2.18.0 Bromo-compounds, Reactions of bromo-compoundsġ2.3.1 Cadmium, Reactions of cadmium sulfate solutionġ2.4.1 Calcium, Reactions of Ca and compoundsġ2.5.0 Chromium, Reactions of chromium compoundsġ2.13.0 Phosphorus, Reactions of phosphorus, Pġ2.17.1 Strontium, Reactions of Sr compoundsġ2.20.1 Tin, Reactions of tin and tin compoundsġ2.21.1 Zn, Reactions of zinc and zinc compoundsġ2.2.2.2 Arsenic, Reactions of As and compoundsġ2.2.2.3 Wood treated with copper chrome arsenate, (CCA)Ĭhemicals Not permitted in schools, Australia: Arsenic compoundsġ2.5.9 Chromium ions in solution, hexaaquachromium ion, Cr(H2O)6 3+ġ2.5.7 Chromic acid, ionization reaction, H2CrO4ġ2.5.5 Oxidize chromium compounds to chromates, CrO4 2-ġ2.5.6 Prepare potassium dichromate, K2Cr2O7ġ2.5.1 Reactions of chromium, Cr, and chromium compoundsġ2.5.3 Reactions of dichromates, Cr2O7 2-, potassium dichromateġ2.13.1 Prepare microcosmic salt, Na.NH4.H.PO4.4H2Oġ2.13.2 Prepare phosphorus trichloride, PCl3ġ2.13.3 Prepare phosphorus pentachloride, PCl5ġ2.13.6 Reactions of phosphorus, P, and phosphates, PO4 3-ġ2.13.8 Phosphorus trichloride with waterġ2.15.1 Prepare silicic acid and pure silicaġ2.15.2 Prepare silica, SiO2, and silicon, Siġ2.15.5 Prepare silicon glass, coloured glassġ2.15.6 Prepare silicon glass in a furnaceġ2.15.8 Silicon reverse-resistance temperature effectġ2.15.9 Silly putty, silicone, bouncing putty (Dow Corning 3179 dilatant compound) "Tricky Putty"ġ2.18.2 Bromoacetic acid, bromoethanoic acidġ2.18.5 Bromochloromethane, methylene chlorobromideĥ.0 Bromocresol green, (acid-base indicator)ħ.0 Bromophenol blue, (acid-base indicator) SbOCl (s) + 2HCl.ģ. Reactions of metallic elements and compounds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
